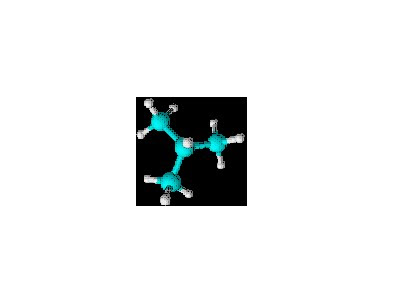
As promised, here are some right out of the textbook.
The first one is in condensed notation: CH
3CH
2CH(CH
3)CH
2CH
3Since I am so good, I immediately recognize that the third carbon has a one-carbon branch. Other than that, this is a straight chain. But let's not get ahead of ourselves. We are doing this the right way. We start with the last part of the name. With no heteroatoms, this is a hydrocarbon. With no multiple bonds, it's an alkane. With no rings, it's an acyclic alkane. We know the name must end in "-ane." Next, what's the longest carbon chain? Five. So the parent name is pentane. Branches? Yes, at the third carbon (counting either way). And the branch is a methyl group. Therefore, the name of this compound is...
...3-methylpentane. And you know what else? I checked the answer in the study guide and I was right! Woo hoo, Stephen got something right. Anyway...
(CH
3)
3CCH
2CH(CH
2CH
3)
2This one is harder. First we have three methyl groups attached to one carbon. That carbon links to another that links to another, which is attached to two ethyl groups. Which methyl group and which ethyl group is considered part of the longest carbon chain does not matter because the groups are identical (that is, the methyl groups are identical to each other and the ethyl groups are identical to each other). So adding those three carbons to the rest of the chain, we find that the longest carbon chain is six carbons long, so this is a hexane.
Which group gets priority? In this case, we go in alphabetical order. "E" comes before "M." So this should be...
...3-ethyl-5,5-dimethylhexane. Or not. Oops. I started at the wrong end. It's actually 4-ethyl-2,2-dimethylhexane. It's that instead of the one I thought it was because 2 is lower than 5. It doesn't matter that 3 is lower than 4 because the method that gives the lowest number period is the one that gets priority, not the one that gives the lowest sum or anything like that. I hope you learned your lesson. Moving on.
CH
3(CH
2)
3CH(CH
2CH
2CH
3)CH(CH
3)
2A propyl group? No, that's part of the longest carbon chain. They're trying to trick us. Starting from the left we have a carbon and then a string of three more, so that's four in a row. Then there's another (five) with that propyl group branching off. If we count going up the propyl group we get three more (eight). If we treat the propyl group as a branch, we get another carbon with two methyl groups, one of which would be a branch, making the total length seven. Sneaky textbook. This is actually an octane.
If we start from the end of what's being labeled as a propyl group (but is actually part of the chain) we get a branch at the fourth carbon. Starting from the left makes it at the fifth, so we start from the end of the propyl group instead. The branch consists of three carbons and two of them are attached to the other, which is where the branch connects, so it's an isopropyl group, meaning the compound is...
...4-isopropyloctane. And I'm right. I rule.
Enough of these condensed structures!

I used MS Paint because it was a small one and it's kind of hard to make them look less awful on ChemSketch. Anyway, this one seems easy to me. Five carbons long means pentane. Two methyl branches at the second carbon and two at the fourth. Therefore...
...2,2,4,4-tetramethylpentane. And I am right again. Excellent.

It's seven carbons long, but there are a couple of different ways to arrive at that. The one that give the lowest number to a branch is the one that simply starts on the far left, for a methyl group at the second carbon. There's another one at the fifth carbon and an ethyl group at the third, so this is...
...3-ethyl-2,5-dimethylheptane. And I'm right yet again. Three in a row! Let's do one more.

I moved back to MS Paint again when I perhaps should not have. But ChemSketch was being annoying (it kept trying to put rings into this). Obviously this one is larger than the other ones so far, but the principle is the same. The longest carbon chain is ten. The fastest we can get to a branch with it is on the second carbon, again counting from the far left. From there we label the other branches and put them in the proper order. About that, the branch on the fifth carbon is a
sec-butyl group. When alphabetizing the branch names, this is treated as a "B" and not as an "S." The same would be true for
tert-butyl but
not for isobutyl. Unnecessarily confusing, I know. But in this case it does slightly affect the name, which is...
...5-
sec-butyl-3-ethyl-2,7-dimethyldecane. And that's pretty much all there is to it. The study guide I used to check my answers breaks the process into three steps.
- Name the parent chain by finding the longest C chain.
- Number the chain so that the first substituent gets the lower number. Then name and number all substituents, giving like substituents a prefix (di, tri, etc.).
- Combine all parts, alphabetizing the substituents, ignoring all prefixes except iso.
It takes some getting used to, but this is the basis for how other compounds, even ones with multiple functional groups, are named.
 It's a ring made of six carbons, so it's a cyclohexane. Only one of the positions in the ring has any groups attached to it, and both groups are methyl groups, so it's 1,1-dimethylcyclohexane.
It's a ring made of six carbons, so it's a cyclohexane. Only one of the positions in the ring has any groups attached to it, and both groups are methyl groups, so it's 1,1-dimethylcyclohexane. Another cyclohexane, obviously. This one has two groups at two different ring positions. The positions are across from each other in a 1,4 relationship. But which group gets numbered "1" and which one gets numbered "4"? Well, one is a methyl group and the other is a butyl group (four carbons in a straight chain). Alphabetical order determines which one comes first, so this is 1-butyl-4-methylcyclohexane.
Another cyclohexane, obviously. This one has two groups at two different ring positions. The positions are across from each other in a 1,4 relationship. But which group gets numbered "1" and which one gets numbered "4"? Well, one is a methyl group and the other is a butyl group (four carbons in a straight chain). Alphabetical order determines which one comes first, so this is 1-butyl-4-methylcyclohexane. The chain is bigger than the ring this time. So this compound is, as far as naming goes, defined as a five-carbon chain with a group attached at the first carbon, and the group that is attached is a cyclopropane ring. Therefore, we have 1-cyclopropylpentane.
The chain is bigger than the ring this time. So this compound is, as far as naming goes, defined as a five-carbon chain with a group attached at the first carbon, and the group that is attached is a cyclopropane ring. Therefore, we have 1-cyclopropylpentane. The ring is a cyclopentane. Three groups this time, and all of them methyl groups, which makes naming this easy. Almost so easy that you could do it by yourself. But how do we number these groups? It doesn't matter which way we count, the smallest number we can start with is 1. This compound is 1,2,3-trimethylpentane.
The ring is a cyclopentane. Three groups this time, and all of them methyl groups, which makes naming this easy. Almost so easy that you could do it by yourself. But how do we number these groups? It doesn't matter which way we count, the smallest number we can start with is 1. This compound is 1,2,3-trimethylpentane. This one is trickier. We definitely have a cyclohexane ring, but what are those groups attached to it. Well, let's start with the smaller one. It's an isopropyl group. See that? Probably not. Well, I told you, so now you know. Isopropyl group. The other one has four carbons. You might remember that there are four such groups possible. And if you have really been paying attention, it's clear that this is a sec-butyl group. Alphabetical order again, but the only prefix that matters for that is "iso-." That means the sec-butyl group is first. So this compound is 1-sec-butyl-2-isopropylcyclohexane.
This one is trickier. We definitely have a cyclohexane ring, but what are those groups attached to it. Well, let's start with the smaller one. It's an isopropyl group. See that? Probably not. Well, I told you, so now you know. Isopropyl group. The other one has four carbons. You might remember that there are four such groups possible. And if you have really been paying attention, it's clear that this is a sec-butyl group. Alphabetical order again, but the only prefix that matters for that is "iso-." That means the sec-butyl group is first. So this compound is 1-sec-butyl-2-isopropylcyclohexane.